Why PLA Production Is Growing Globally
PLA accounted for approximately 26 percent of global bioplastics demand in 2022, making it the most widely consumed bio-based polymer. Its adoption has expanded across packaging, additive manufacturing, medical devices, and agricultural applications.
This growth is primarily driven by regulatory pressure on fossil-based plastics, carbon reduction targets, and supply chain diversification. PLA, derived from renewable carbohydrates such as corn, sugarcane, and cassava, provides a scalable alternative with defined processing behavior.
For new entrants, the constraint is not material chemistry but system execution. Production stability, product consistency, and cost control depend on the integration of reaction, purification, and polymerization stages within a single coordinated system.
What a Turnkey PLA Production System Includes
A turnkey PLA production system covers the full process chain from lactic acid to polymer-grade PLA, delivered as an operational facility with defined performance targets.
The scope typically includes:
- Process design and simulation
- Core equipment supply – reactors, crystallizers, distillation systems
- Automation and control systems
- Installation, commissioning, and performance validation
System integration determines overall performance. Individual unit operations must operate under coordinated thermal, pressure, and material balance conditions to maintain product consistency and throughput.
In practice, project risk is reduced when process design, equipment selection, and commissioning are handled within a unified engineering framework rather than fragmented across multiple suppliers.
Main PLA Production Routes Explained
PLA production follows two industrial pathways, differentiated by polymer formation method.
Direct Polycondensation
Lactic acid undergoes esterification, generating water as a byproduct. Continuous water removal under vacuum shifts the equilibrium.
Observed constraints include:
- Molecular weight typically below 130 kDa
- Slow reaction kinetics
- Residual monomers and oligomers
Solid-state polymerization can increase molecular weight, but adds process complexity and longer cycle time.
Lactide Route via Ring-Opening Polymerization
The lactide route remains the dominant industrial pathway due to control over molecular structure and product performance.
Process sequence:
- Lactic acid pre-polymerization to oligomers
- Depolymerization to lactide
- Lactide purification
- Ring-opening polymerization to PLA
This route enables higher molecular weight, controlled stereochemistry, and consistent thermal properties. Industrial plants targeting stable output and scalable capacity typically adopt this configuration.
From Biomass to Lactic Acid
PLA production begins with carbohydrate feedstocks. Sugars derived from corn, sugarcane, or cassava are converted into lactic acid through microbial fermentation.
The industrial sequence includes:
- Starch hydrolysis to fermentable sugars
- Microbial fermentation
- Acidification and filtration
- Esterification and distillation
While fermentation defines upstream yield, downstream purification determines polymerization performance. Impurities introduced at this stage propagate through lactide formation and affect final product quality.
Lactide Production and Purification
Lactide is the critical intermediate that determines polymer structure and performance.
Lactide Formation
Low molecular weight oligomers are thermally depolymerized at temperatures between 200 and 250 degrees Celsius under high vacuum conditions. The reaction produces lactide vapor, which is condensed into crude lactide.
Purification System
High-purity lactide is required for controlled polymerization.
Typical separation steps include:
- Distillation to remove water, residual lactic acid, and oligomers
- Melt crystallization to separate stereoisomers
Optical purity directly affects crystallinity, melting temperature, and mechanical strength. Industrial systems targeting high-performance PLA typically require lactide purity above 99.5 percent.
In practice, crystallization design becomes the primary determinant of impurity removal efficiency. Systems requiring high optical purity rely on precise temperature control and crystal growth management, capabilities commonly associated with high-purity intermediate separation processes.
How PLA Polymerization Works
PLA is produced through ring-opening polymerization under controlled reactor conditions.
Reaction Conditions
- Temperature range: 180 to 220 degrees Celsius
- Inert atmosphere to prevent oxidation
- Metal-based catalysts
- Reaction time between two and four hours
Reactor Design
As polymer chains grow, melt viscosity increases significantly, creating constraints in mixing and heat transfer.
Common reactor configurations include:
- High-torque stirred reactors
- Horizontal reactors with enhanced heat transfer
- Continuous twin-screw systems
Polymer properties are determined by:
- Catalyst concentration
- Monomer-to-initiator ratio
- Residence time
Temperature uniformity directly affects stereochemistry and degradation. Reactor performance therefore becomes a limiting factor in scale-up and product consistency.
Batch vs Continuous Production Systems
Production configuration determines capital efficiency and operational stability.
Batch Systems
- Flexible operation and product variation
- Easier process control
- Higher downtime and labor requirements
Continuous Systems
- Stable product quality under steady-state operation
- Higher throughput at industrial scale
- Greater initial capital investment
Continuous systems are typically adopted at capacities above 50,000 tons per year. Their advantage lies in consistent residence time distribution and improved devolatilization efficiency.
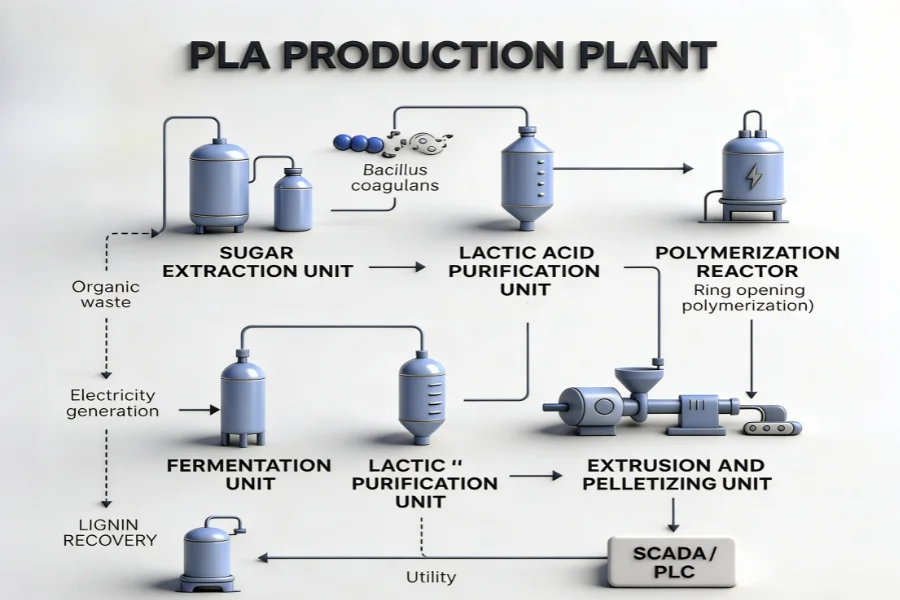
How a PLA Production Plant Is Structured
A PLA production plant operates as an integrated system rather than a sequence of independent units.
Core Process Flow
Feedstock → Lactic acid → Pre-polymerization → Lactide formation → Lactide purification → Polymerization → Devolatilization → Pelletizing
Supporting Systems
- Vacuum systems for water and monomer removal
- Heat integration for energy efficiency
- Solvent recovery and recycling
- Automated control systems
System-level material and energy balances determine plant performance. Local optimization at individual units is insufficient if integration across the process chain is not maintained.
Key Challenges in PLA Production and How to Solve Them
Optical Purity Control
Racemization during lactide formation reduces crystallinity and thermal performance.
Mitigation requires:
- Tight temperature control
- Advanced crystallization techniques
Heat and Mass Transfer Limitations
Polymerization is exothermic and involves increasing viscosity.
Observed risks include:
- Local overheating
- Broad molecular weight distribution
Effective reactor design improves heat removal and mixing, maintaining stable reaction conditions.
Residual Monomer Removal
Unreacted lactide affects thermal stability and long-term performance.
Industrial solutions include:
- Vacuum devolatilization
- Thin-film evaporation
- Twin-screw degassing
Residual monomer levels below 0.1 percent are typically required for commercial-grade material.
PLA Plant Cost and Scale Planning
PLA production economics are driven by feedstock cost, process efficiency, and plant scale.
Key observations:
- Lactic acid contributes approximately 50 to 60 percent of total production cost
- Energy consumption varies by process route
- Recovery efficiency directly affects yield
Typical benchmarks include:
- Lactic acid consumption between 1.1 and 1.2 tons per ton of PLA
- Initial plant capacity between 10,000 and 50,000 tons per year
- Cost efficiency improves significantly beyond 100,000 tons per year
Production stability and downtime reduction have a direct impact on return on investment. These factors are primarily influenced by system integration and equipment performance rather than raw process selection.
PLA Recycling and End-of-Life Options
PLA is classified as a bio-based polymer with defined end-of-life pathways.
Key routes include:
- Industrial composting under controlled temperature and humidity
- Chemical recycling through hydrolysis or alcoholysis
- Energy recovery through incineration
Degradation performance depends on environmental conditions. Industrial composting remains the most effective route for material breakdown.
Conclusion
PLA production performance is determined by system integration rather than individual process steps. The transition from lactic acid to polymer requires coordinated control across reaction, purification, and polymerization stages.
Turnkey solutions align these stages within a single engineering framework, reducing variability and improving operational stability. Project outcomes depend on the ability to manage high-purity intermediates, control high-viscosity reactions, and maintain system-level balance across the process chain.
In industrial practice, these capabilities are typically associated with engineering teams experienced in complex reaction systems and high-purity separation processes.
FAQ
What is the primary raw material for PLA production
PLA production is based on lactic acid derived from fermentable sugars such as corn, sugarcane, or cassava. Feedstock selection directly affects cost structure, regional supply stability, and long-term production economics, making it a key consideration during project planning.
Which process route is preferred for industrial PLA production
The lactide route using ring-opening polymerization is the dominant industrial method. It provides better control over molecular weight, stereochemistry, and product consistency compared to direct polycondensation, making it more suitable for large-scale, high-performance PLA production.
Why is lactide purification critical
Lactide purity determines polymer stereochemistry, molecular weight distribution, and thermal stability. Impurities such as meso-lactide or residual oligomers can reduce crystallinity and degrade performance, making purification a critical step in achieving consistent industrial-grade PLA.