From Molecular Properties to Industrial Piping Design Challenges
Hydrogen cyanide (HCN) is a highly toxic and volatile intermediate used in acrylonitrile, adiponitrile, and fine chemical synthesis. Its physical properties create elevated containment requirements in industrial piping systems.
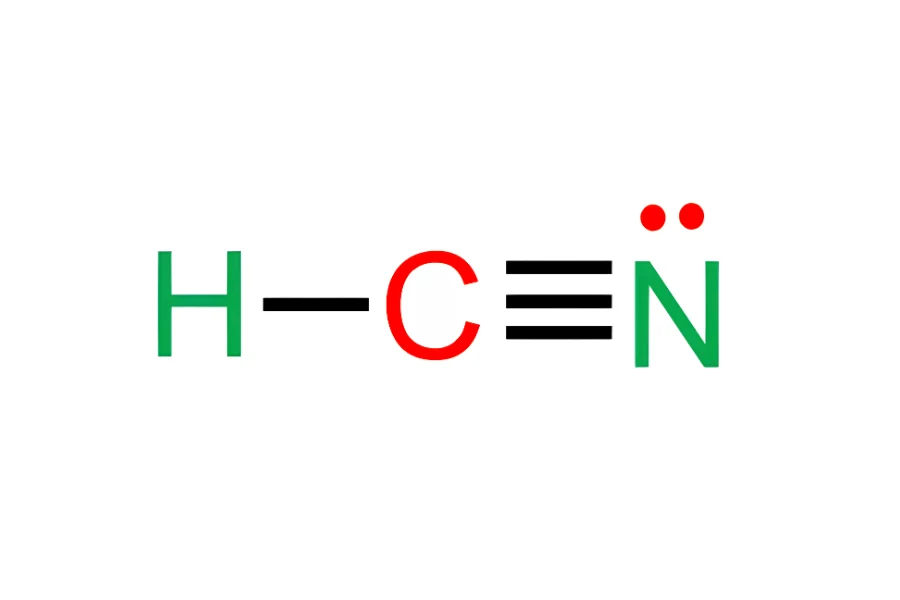
HCN has a molecular weight of 27.025 g/mol, a linear structure (H–C≡N), and a boiling point of approximately 26 °C. At ambient conditions, it exists close to the liquid–gas phase boundary, increasing vapor generation sensitivity.
In piping systems, HCN service introduces elevated risks of fugitive emissions due to its low viscosity, high diffusion rate, and broad flammability range. Conventional utility piping designs are generally insufficient for long-term containment.
Safe handling requires integrated containment design, including welded piping systems, bellows sealed valves, controlled monitoring, and engineered emergency isolation strategies.
How HCN Molecular Properties Influence Piping Integrity
Low Molecular Weight and Diffusion Behavior
HCN exhibits high molecular mobility and low viscosity. These properties increase diffusion through microscopic sealing defects.

Leakage risk is typically associated with:
- Valve stem packing interfaces
- Flange gasket surfaces
- Threaded mechanical joints
- Instrument connection points
Containment performance depends on sealing integrity rather than bulk material strength.
Molecular Polarity and Surface Interaction
HCN is a polar molecule with a dipole moment of approximately 2.98 D. This influences adsorption behavior on metallic and polymer surfaces.
Moisture presence may amplify localized compatibility issues in selected materials, particularly under long-term exposure conditions.
Acid-Base Equilibrium and Cyanide Formation
In aqueous systems, HCN partially dissociates into CN⁻ with a pKa of 9.21. Acidic conditions shift equilibrium toward vapor-phase release.
Process systems typically require controlled alkaline conditions to reduce volatilization risk during handling and transfer.
Vapor Pressure and Phase Instability
HCN has a boiling point near ambient temperature, resulting in continuous vapor-liquid phase sensitivity.
Engineering considerations include:
- Flash vapor formation risk
- Pressure fluctuation control
- Venting system design
- Thermal stability management
Small temperature changes can significantly affect vapor concentration.
Flammability and Ignition Sensitivity
HCN has a flammable range of 5.6% to 40% in air. This creates a broad ignition envelope in confined systems.
Key ignition control requirements include:
- Static grounding and bonding
- Electrical classification compliance
- Hot surface control
- Explosion risk mitigation
Primary Failure Modes in HCN Piping Systems
Fugitive Emission Pathways
Long-term leakage typically occurs at sealing interfaces rather than pipe body failure. Even low-level emissions may accumulate to exceed occupational exposure limits.
Packed valve systems may exhibit gradual stem leakage under thermal cycling conditions.
Material Compatibility Constraints
Material selection must account for chemical stability and long-term exposure behavior.
Commonly applied materials include:
- 316L stainless steel
- 304 stainless steel
- Carbon steel under controlled conditions
- PTFE-lined components
Materials generally avoided:
- Copper alloys
- Brass and bronze
- Cast iron
Thermal and Storage Instability
HCN may undergo polymerization under unfavorable conditions, particularly in stagnant or temperature-unstable environments.
Engineering design should minimize:
- Dead legs
- Long residence time zones
- Local overheating regions
- Static liquid accumulation
Toxic Exposure Mechanism
HCN inhibits cytochrome c oxidase, preventing cellular oxygen utilization. Occupational exposure limits are typically set at 10 ppm (OSHA) with 50 ppm defined as IDLH conditions.
Odor detection is not a reliable safety control method due to variable human sensitivity.
Material Selection for HCN Piping Systems
Qualified Structural Materials
Material selection prioritizes chemical stability and permeability resistance.
Typical selections:
- 316L stainless steel for primary process lines
- 304 stainless steel for secondary systems
- Carbon steel with controlled environment management
- PTFE-lined flexible elements
- Seamless pipe configurations
Sealing and Gasket Systems
Seal integrity is critical due to diffusion-driven leakage behavior.
Recommended sealing systems include:
- Spiral wound graphite gaskets with metal reinforcement
- PTFE gaskets under controlled compression
- Metal-seated sealing systems for critical joints
Elastomeric seals are generally limited in continuous HCN service.
Valve and Welding Design Requirements
Bellows Sealed Valve Systems
Bellows sealed valves isolate process fluid from atmospheric exposure at the stem interface.
Engineering benefits include:
- Elimination of stem leakage pathways
- Reduced fugitive emission probability
- Improved long-term sealing stability
- Compliance with ISO 15848 standards
Fully Welded Piping Architecture
Welded systems are preferred for minimizing leakage interfaces.
Typical design features:
- GTAW welded pipe assemblies
- Reduced flange count design
- Radiographic weld inspection
- Post-weld surface passivation
Flanged joints are minimized and used only for maintenance access points.
System-Level Safety Controls
Additional safeguards may include:
- Grounding and bonding systems
- Nitrogen purging
- Pressure relief devices
- Rupture disk protection
- Emergency shutdown interlocks
Leak Detection and Monitoring Systems
Fixed Gas Detection Networks
Electrochemical sensors are deployed at high-risk locations including valves, pumps, and loading systems.
Detection ranges typically cover sub-ppm to tens of ppm levels.
Cross-sensitivity with gases such as SO₂ and NO₂ requires compensation algorithms.
Personal Monitoring Systems
Portable detectors are required for personnel in HCN handling areas. Alarm thresholds are configured below occupational exposure limits.
Integrated Control and SCADA Systems
Gas detection systems are typically integrated into DCS or SCADA platforms.
Automated responses may include:
- Valve isolation
- Ventilation activation
- Alarm signaling
- Emergency shutdown
Emergency Response and Neutralization
Localized Leak Control
Small-scale releases are managed through:
- Immediate isolation
- Nitrogen purging
- Alkaline neutralization
- Absorbent containment
Large-Scale Release Response
Major incidents require:
- Area evacuation
- Upwind personnel relocation
- Ignition source control
- Vapor dispersion management
Medical Intervention
Acute exposure treatment includes:
- 100% oxygen administration
- Hydroxocobalamin
- Sodium nitrite
- Sodium thiosulfate
Rapid response is critical due to fast systemic toxicity onset.
Engineering Support for HCN-Related API Manufacturing
HCN is commonly used in Strecker synthesis, cyanohydrin formation, and nitrile intermediate production in pharmaceutical manufacturing.
Process development requires integrated engineering across:
- Reaction containment design
- Crystallization control systems
- High-toxicity process isolation
- cGMP-aligned manufacturing integration
DODGEN supports engineering design frameworks for hazardous chemical process systems with emphasis on containment integrity, reaction stability, and scalable industrial implementation.
Conclusion
HCN exhibits low molecular weight, high volatility, and broad flammability characteristics that directly influence industrial piping design requirements.
Safe operation depends on integrated engineering systems including welded piping architecture, bellows sealed valves, material compatibility control, and continuous gas monitoring infrastructure.
Industrial containment performance is determined by system integration rather than individual component selection, requiring coordinated design across materials, sealing strategy, and emergency response systems.
FAQ
What is hydrogen cyanide used for in industry?
Hydrogen cyanide is used as a key intermediate in acrylonitrile, adiponitrile, pesticides, and pharmaceutical synthesis. It is widely applied in polymer production and fine chemical manufacturing processes.
Why is hydrogen cyanide so toxic?
HCN blocks cytochrome c oxidase in mitochondria, preventing cells from using oxygen. This leads to rapid cellular hypoxia even when oxygen is available in the bloodstream.
At what concentration (ppm) is hydrogen cyanide toxic?
Hydrogen cyanide becomes hazardous at very low concentrations. Exposure above 10 ppm is regulated in workplaces, while around 50 ppm is considered immediately dangerous to life and health.
What are the key chemical properties of hydrogen cyanide?
HCN is a highly volatile, low-viscosity, polar molecule with a boiling point near 26°C. It is flammable, weakly acidic, and highly diffusive, which increases leakage risk in industrial systems.




