System Characteristics of Hydrocyanic Acid Service
Ácido cianhídrico (HCN) is a weak acid with pKa ≈ 9.2 and limited dissociation in aqueous systems. Under dry, high-purity conditions at temperatures near ambient, general corrosion rates on common alloys remain low.
In industrial operation, HCN is rarely present as a pure system. It is volatile at near-ambient temperature, highly toxic at low ppm levels, and sensitive to trace impurities. As a result, equipment integrity is governed by impurity-driven corrosion and containment reliability rather than nominal acid strength.
Baseline Corrosion Behavior Under Controlled Conditions
Under dry conditions with impurity levels below detection limits:
- Austenitic stainless steels such as 316L typically exhibit corrosion rates below 0.01 mm/year at temperatures below 25°C
- Passive film stability is maintained in the absence of oxidizing or halide species
- Uniform corrosion remains the dominant mechanism
These conditions are not stable in most process environments and should not be used as the basis for material selection.
Impurity-Driven Corrosion – Dominant Engineering Constraint
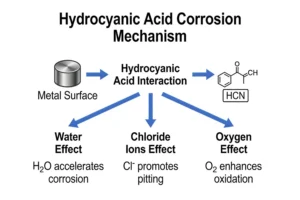
Corrosion behavior in HCN systems is governed by the presence of water, halides, oxidants, and acidic contaminants. These factors alter both electrochemical conditions and material stability.
Water Content
Water concentrations above approximately 0.5–1.0 wt% introduce hydrolysis and secondary reactions. Observed effects include:
- Formation of acidic or basic byproducts
- pH variation across the system
- Increase in corrosion rates of stainless steels to 0.05–0.20 mm/year depending on temperature
Halide Contamination
Chloride concentrations as low as 10–50 ppm are sufficient to initiate localized corrosion in austenitic stainless steels under tensile stress.
- Pitting potential decreases significantly in the presence of Cl⁻
- Stress corrosion cracking has been observed in 304 and 316 grades at temperatures above 20°C
- Localized penetration can occur without measurable uniform corrosion
Oxidizing Species
Trace oxygen or oxidizing acids disrupt passive films:
- Passive breakdown potential decreases
- Transition from passive to active corrosion occurs
- Localized corrosion rates exceed 0.5 mm/year in affected regions
Acidic Contaminants
The presence of HCl or H₂SO₄ shifts the system from weakly acidic to strongly acidic behavior:
- Corrosion rates increase by an order of magnitude relative to pure HCN
- Uniform corrosion becomes significant in addition to localized attack
Corrosion Mechanism Model
Corrosion in HCN systems follows a conditional transition model:
- Base state – low corrosion under dry, impurity-free conditions
- Trigger state – introduction of water, halides, or oxidants
- Failure state – localized corrosion, pitting, crevice corrosion, and stress corrosion cracking
Material selection must be based on the trigger and failure states rather than the base state.
Typical Failure Modes in Pumps and Valves
| Modo de fallo | Trigger Condition | Observed Impact |
|---|---|---|
| Stress corrosion cracking | Cl⁻ ≥ 20 ppm with residual stress | Rapid crack propagation and loss of containment |
| Pitting corrosion | Local chloride concentration | Penetration without warning |
| Crevice corrosion | Flange and seal interfaces | Accelerated localized attack |
| Elastomer degradation | Chemical incompatibility | Loss of sealing pressure and leakage |
Failure progression is typically non-linear and may lead to sudden release rather than gradual degradation.
Material Selection Framework
Selection Criteria
Material selection must be based on:
- Full chemical composition including trace impurities
- Temperature and pressure envelope
- Flow velocity and erosion potential
- Operating mode including startup and upset conditions
Lifecycle cost must include maintenance intervals, failure probability, and safety impact.
Metallic Materials
Austenitic Stainless Steels
- 304 / 304L
Acceptable only for dry, high-purity HCN with impurity levels below critical thresholds
Failure risk increases significantly above 10 ppm chloride - 316 / 316L
Baseline material for controlled systems
Corrosion rates remain below 0.05 mm/year in low-impurity conditions
Risk of localized corrosion persists in the presence of halides - Stabilized grades (321)
Improved resistance to intergranular corrosion in welded assemblies
Duplex Stainless Steels
Duplex alloys such as 2205 exhibit improved resistance to stress corrosion cracking. However:
- Pitting resistance remains dependent on PREN value
- Performance is not stable in mixed impurity systems without validation
Nickel-Based Alloys
- Hastelloy C-276
Maintains corrosion rates below 0.01 mm/year in mixed impurity systems including halides
Resistant to pitting, crevice corrosion, and stress corrosion cracking
Considered a primary material for high-risk HCN service - Monel 400
Compatible with HCN systems but typically selected based on specific process requirements - Inconel 625
Provides stable performance in mixed acid environments but is selected selectively due to cost
Titanium Alloys
Titanium Grade 2 and Grade 7 demonstrate stable performance in:
- Oxygen-containing systems
- Low-halide environments
However:
- Fluoride concentrations above trace levels result in rapid degradation
- Application requires impurity control and validation testing
Non-Metallic and Lined Materials
Fluoropolymers
- PTFE
Chemically inert across all typical HCN compositions
No measurable corrosion under standard conditions - PFA / FEP
Similar chemical resistance with improved manufacturability - PVDF
Mechanical strength advantages with acceptable chemical resistance
Limitations include:
- Reduced resistance to abrasion
- Limited structural strength
- Sensitivity to vacuum-induced deformation
Sealing Systems – Primary Containment Risk
Seal failure represents the highest probability of release in HCN systems.
| Material | Performance | Notas |
|---|---|---|
| FKM (Viton) | Stable | Standard sealing material |
| PTFE-based seals | Highly stable | Preferred for high-integrity systems |
| NBR | Conditional | Limited use in non-critical service |
| EPDM | Unstable | Rapid swelling and degradation |
Seal selection must consider both chemical compatibility and temperature-dependent mechanical properties.
Pump and Valve Configuration
Pump Systems
- Centrifugal pumps require minimum flow control to prevent thermal damage to seals
- Magnetic drive and canned motor pumps eliminate dynamic seals and reduce leakage probability
- Diaphragm pumps provide high containment integrity for low-flow applications
Valve Systems
- Bellows-sealed valves eliminate stem leakage paths
- Lined valves provide chemical isolation in aggressive environments
- Ball valves require antistatic design to mitigate electrostatic discharge risk
Threaded connections are not recommended due to crevice formation and incomplete decontamination.
Operating Envelope Control
- Temperature maintained below 20°C to limit volatility and reaction kinetics
- Flow velocity controlled to reduce erosion and electrostatic accumulation
At temperatures below −10°C, elastomer flexibility decreases and sealing force must be verified.
Safety Design and Maintenance
Design Measures
- Dual sealing systems with pressure-monitored barrier fluids
- Inert gas purging systems for isolation and maintenance
- Grounding of all conductive components
- High-integrity gasket systems for flange connections
Maintenance Practices
- Scheduled seal replacement based on operating hours rather than failure
- Full decontamination using inert gas and chemical neutralization prior to maintenance
- Thickness monitoring at high-risk locations including elbows and valve seats
Failure to maintain sealing integrity is the primary cause of hazardous release.
Failure Scenario Analysis
Observed failure scenarios in HCN systems include:
- Austenitic stainless steel exposed to 20–50 ppm chloride leading to stress corrosion cracking within months of operation
- Elastomer seal swelling resulting in loss of sealing force and leakage
- Lined components failing under vacuum or abrasive conditions
These scenarios demonstrate that failure is driven by localized conditions rather than average system chemistry.
Integrated Engineering Approach
Material performance in HCN service is determined by the interaction between process chemistry, operating conditions, and equipment design.
DODGEN applies a process-integrated approach in API synthesis and crystallization systems involving HCN. Material selection is defined in relation to reaction composition, impurity evolution, and separation conditions, rather than nominal chemical identity.
This approach reduces dependence on conservative material selection and improves predictability of equipment performance over the operating lifecycle.
Conclusión
Hydrocyanic acid systems are defined by impurity sensitivity and containment risk rather than intrinsic acid strength. Material selection for pumps and valves must be based on worst-case conditions, including trace impurities and process variability.
Reliable operation requires alignment between material capability, process control, and sealing integrity. A structured, data-based approach to selection and design is necessary to ensure both safety and long-term performance.
PREGUNTAS FRECUENTES
Why is HCN not considered a strong acid?
HCN is classified as a weak acid because it only partially dissociates in aqueous solution, with a dissociation constant of approximately 6.2 × 10⁻¹⁰. The strong carbon–hydrogen bond limits proton release. In industrial systems, its corrosion behavior is not determined by acidity, but by impurities such as water, halides, and oxidizing species.
Is HCN or HCl stronger?
Hydrochloric acid is significantly stronger than HCN. HCl dissociates almost completely in water, while HCN remains largely undissociated. Despite this, HCN systems can present higher engineering risk due to toxicity, volatility, and impurity-driven corrosion mechanisms that are not directly related to acid strength.
Why is hydrogen cyanide highly toxic?
Hydrogen cyanide inhibits cellular respiration by binding to cytochrome c oxidase in mitochondria, preventing oxygen utilization at the cellular level. Exposure at concentrations above approximately 50 ppm can lead to rapid physiological failure. Toxicity, rather than acidity, defines the primary hazard in HCN processing environments.
Is HCN more acidic than water?
HCN is more acidic than water, as indicated by its lower pKa value. However, both substances are weak proton donors. In practical systems, this difference has limited relevance to corrosion behavior, which is governed by impurity composition, temperature, and process conditions rather than intrinsic acid strength.