Medical Grade PGA Suture Performance Requirements
Polyglycolic acid (PGA) is widely used in absorbable surgical sutures due to its predictable hydrolysis behavior, mechanical strength retention profile, and low tissue reactivity. Applications include general surgery, gynecology, ophthalmic procedures, gastrointestinal procedures, and soft tissue approximation.
In medical applications, performance requirements are defined by time-dependent mechanical stability and controlled degradation behavior inside biological environments.
Key performance parameters include:
- Absorption window of approximately 60 to 90 days
- Tensile strength retention during early wound healing phase
- Controlled hydrolysis kinetics under physiological conditions
- Low inflammatory tissue response
- Stable knot security under dynamic loading conditions
These requirements define strict constraints across the entire manufacturing chain, from polymer synthesis to final sterilized packaging.
Production consistency is determined not only by raw material quality, but also by the stability of each process stage and equipment integration across the system.
Technical Constraints in Medical Grade PGA Manufacturing
Medical grade PGA production is sensitive to both thermal degradation and moisture-induced chain scission during synthesis and processing. Molecular weight distribution stability directly influences extrusion behavior, fiber formation, and long-term degradation performance.
Inconsistent polymer characteristics may lead to:
- Unstable melt viscosity during extrusion
- Reduced filament tensile strength
- Irregular hydrolysis behavior
- Variability in absorption timing
As a result, process stability requirements extend beyond individual equipment performance and must be managed at the system level.
Manufacturing reliability depends on controlled interaction between polymerization, fiber formation, braiding, coating, and sterilization processes.
Polymerization System Requirements for High Molecular Weight PGA
Vacuum Reactor System Design
High molecular weight PGA production relies on controlled polymerization environments designed to maintain chain growth stability while minimizing degradation reactions.
Vacuum reactor systems are typically designed to support:
- Operating vacuum levels below 1 mbar
- Precise temperature stability within ±1°C
- Continuous removal of condensation byproducts
- Controlled catalyst distribution systems
- Inert gas protection during reaction cycles
Material selection is critical due to corrosive intermediates generated during polymerization. Stainless steel or Hastelloy-based reactor systems are commonly used to ensure long-term operational stability.
Molecular weight consistency at this stage directly determines downstream extrusion stability and fiber mechanical performance.
Process Integration Considerations
Polymer synthesis must be integrated with purification and crystallization control systems to stabilize molecular distribution before fiber processing. Variability at this stage propagates through all downstream mechanical properties of the suture system.
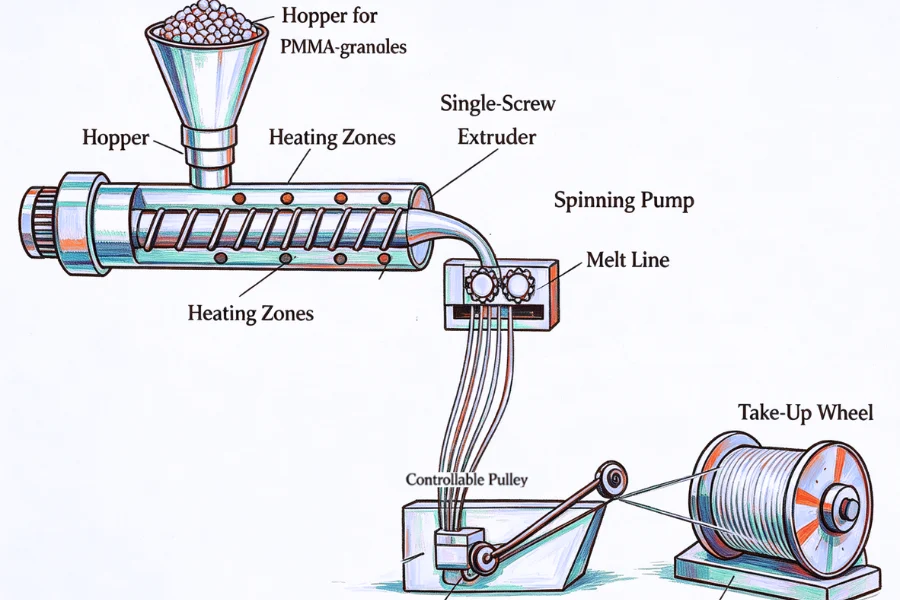
Melt Extrusion and Fiber Spinning System Requirements
Thermal Stability and Filament Formation Control
Melt extrusion is a critical transformation stage where polymer resin is converted into continuous fibers. Process stability directly affects filament morphology and mechanical strength.
Key system requirements include:
- Temperature control accuracy within ±0.5°C
- Uniform spinneret design with micron-level precision
- Controlled cooling systems for solidification stability
- Automated tension regulation during fiber drawing
- Stable melt viscosity management across extrusion cycles
Processing temperatures are typically maintained between 220°C and 240°C to balance flow stability and thermal degradation risk.
Molecular Orientation and Strength Development
Fiber drawing systems introduce molecular orientation, which determines tensile strength retention and elasticity behavior during clinical application.
Inconsistent drawing ratios may lead to:
- Uneven fiber diameter distribution
- Reduced knot security performance
- Variable tensile retention across batches
Inline Process Monitoring Systems
Modern PGA fiber production systems increasingly rely on inline monitoring technologies to ensure continuous process stability:
- Laser-based diameter measurement systems
- Melt pressure feedback loops
- Real-time tension control systems
- Statistical process control (SPC) frameworks
- Automated defect detection modules
These systems reduce process drift and improve batch-to-batch reproducibility, which is critical for medical device compliance.
Braiding System Requirements for Multifilament Structures
Tension-Controlled Braiding Architecture
Most PGA surgical sutures use multifilament braided structures to achieve a balance between flexibility, knot security, and handling performance.
Braiding systems must maintain synchronized tension control across multiple filaments to prevent structural inconsistencies.

Key equipment requirements include:
- Multi-carrier high-speed braiding systems (16–32 carriers)
- Dynamic tension balancing systems
- Adjustable braid angle and pitch control
- Real-time filament break detection
Mechanical Behavior Dependency
Braiding geometry directly influences:
- Knot holding reliability
- Surface flexibility
- Tissue interaction resistance
- Load distribution under tension
- Coating absorption behavior
Downstream coating performance is highly dependent on braid uniformity and structural stability.
Coating System and Surface Engineering Requirements
Controlled Coating Application Systems
PGA sutures are commonly coated with polycaprolactone and calcium stearate systems to optimize surface lubrication and reduce tissue drag.
Coating system requirements include:
- Controlled solution concentration management
- Continuous mixing and homogenization systems
- Multi-zone drying tunnels with thermal gradient control
- Solvent recovery and circulation systems
Functional Coating Parameters
Coating thickness is typically controlled within 5 to 10 microns. Deviations can significantly affect:
- Hydrolysis rate stability
- Tissue friction resistance
- Absorption behavior consistency
- Surface handling performance during surgery
Coating uniformity is directly linked to braid structure stability and fiber surface energy characteristics.
Needle Manufacturing System Requirements
Precision Mechanical and Laser Processing
Surgical needles used in PGA sutures require high precision forming systems to ensure consistent penetration performance and structural integrity.
Core manufacturing systems include:
- Centerless grinding systems for edge precision
- Laser drilling systems for controlled eye formation
- Automated curvature forming systems (1/2, 3/8 circular profiles)
- Surface polishing systems for friction reduction
- Mechanical penetration force validation systems
Material and Structural Stability
Needles are typically manufactured from 300-series stainless steel to ensure corrosion resistance and mechanical strength stability during sterilization and storage.
Performance stability depends on:
- Edge sharpness retention
- Elastic deformation resistance
- Surface defect control
- Post-sterilization structural integrity
Sterilization and Packaging System Requirements
EO Sterilization Process Control
Ethylene oxide (EO) sterilization is widely used due to compatibility with thermally sensitive absorbable polymers.
Process control systems must manage:
- Temperature stability during sterilization cycles
- Humidity and gas concentration control
- Exposure time regulation
- Post-sterilization aeration efficiency
- Residual EO removal validation
Improper sterilization control may affect polymer degradation stability and mechanical retention properties.
Barrier Packaging Systems
Sterile barrier systems are designed to maintain long-term product integrity under storage conditions.
Typical configurations include:
- Aluminum foil composite sterile pouches
- Double-layer sterile packaging systems
- Heat-sealing validation systems
- Cleanroom-integrated packaging lines
Quality Control and Validation Systems
Mechanical and Chemical Validation
Quality control in PGA suture manufacturing requires multi-layer validation across mechanical, chemical, and biological performance parameters.
Key testing systems include:
- Universal tensile testing systems for strength validation
- Knot security testing systems
- Hydrolysis simulation systems using PBS at 37°C
- Gas chromatography for EO residue analysis
- Surface defect detection systems
- Molecular weight distribution analysis
Process Stability Assurance
Validation systems are used not only for final product testing but also for continuous process verification across production stages.
Consistency in mechanical behavior is directly correlated with stability in polymer synthesis and fiber processing systems.
Cleanroom and Regulatory Compliance Systems
Medical PGA suture production is conducted under controlled cleanroom environments typically classified as ISO Class 7 or higher.
Environmental control systems regulate:
- Particulate contamination levels
- Temperature and humidity stability
- Microbial contamination control
- Personnel and material flow management
Regulatory frameworks commonly include:
- ISO 13485 quality management systems
- United States Pharmacopeia (USP) standards
- European Pharmacopoeia (EP) requirements
- Medical device GMP compliance systems
Traceability across the production chain is a critical requirement for regulatory approval and long-term quality assurance.
결론
Medical grade PGA surgical suture manufacturing requires a tightly integrated production system combining polymer synthesis control, precision extrusion, braided fiber engineering, surface coating regulation, sterilization process control, and multi-layer quality validation.
Product performance is determined not by isolated material properties, but by the stability and interaction of each production subsystem within a controlled engineering environment.
As demand for absorbable surgical materials continues to increase, production systems are evolving toward higher levels of automation, process monitoring integration, and molecular-level consistency control across the entire manufacturing chain.
In this context, process stability, crystallization control, and polymer purification system design remain critical factors in achieving reproducible mechanical performance and regulatory compliance in medical-grade PGA suture production.
자주 묻는 질문
What is PGA suture material?
PGA suture is a synthetic absorbable polymer made from polyglycolic acid. It is widely used in surgical procedures due to predictable hydrolysis and reliable tensile strength retention.
Is polyglycolic acid an absorbable suture?
Yes. PGA is a fully absorbable suture material that degrades through hydrolysis in the body, typically within 60 to 90 days depending on molecular structure and environmental conditions.
What is the difference between PGA and PGCL sutures?
PGA absorbs faster and provides higher initial strength, while PGCL offers longer absorption time and greater flexibility. Selection depends on tissue healing requirements and surgical application.
How does PGA suture degrade in the body?
PGA sutures degrade through hydrolysis, where water breaks polymer chains into glycolic acid. The process is controlled by molecular weight, crystallinity, and fiber structure.




